Discovery of Tumor-Specific Irreversible Inhibitors of Stearoyl Coenzyme A Desaturase
Abstract
Targeting metabolic pathways in cancer has emerged as a powerful therapeutic strategy, offering alternatives to traditional cytotoxic treatments. One promising enzyme is Stearoyl-CoA Desaturase (SCD), which is crucial for lipid metabolism and frequently overexpressed in tumors. However, the challenge of selectively inhibiting SCD in cancer cells without affecting normal tissues has limited clinical translation. Recent research has led to the discovery of tumor-specific irreversible SCD inhibitors that covalently bind and inactivate the enzyme selectively within cancer cells. These compounds demonstrate strong anti-tumor effects with minimal toxicity, paving the way for safer and more effective metabolic therapies. This blog series explores the biological rationale, discovery process, mechanism of action, and broader implications of these novel inhibitors in cancer drug development.
A New Era in Cancer Therapeutics: Targeting Lipid Metabolism
Cancer therapy has entered a transformative era, where researchers are going beyond traditional targets and exploring new biological pathways that tumors depend on for growth and survival. One such emerging area is lipid metabolism, a critical process that cancer cells exploit to meet their energy and structural demands. Among the enzymes involved in this process, Stearoyl-CoA Desaturase (SCD) has gained significant attention.
SCD is an endoplasmic reticulum-associated enzyme responsible for converting saturated fatty acids into monounsaturated fatty acids—key building blocks of membrane lipids, signaling molecules, and energy stores. Its role becomes particularly crucial in rapidly proliferating cancer cells that require a steady supply of lipids to support their aggressive growth. Studies have shown that many tumors exhibit overexpression of SCD, which is linked to cancer progression, drug resistance, and poor prognosis.
However, targeting metabolic enzymes like SCD poses a challenge: these enzymes are also vital for normal cells. Thus, broad-spectrum inhibition can result in significant toxicity. This has led scientists to pursue a new goal—tumor-specific inhibition of metabolic targets. By identifying compounds that selectively inhibit SCD in cancer cells while sparing healthy tissues, researchers aim to develop safer and more effective therapeutics.
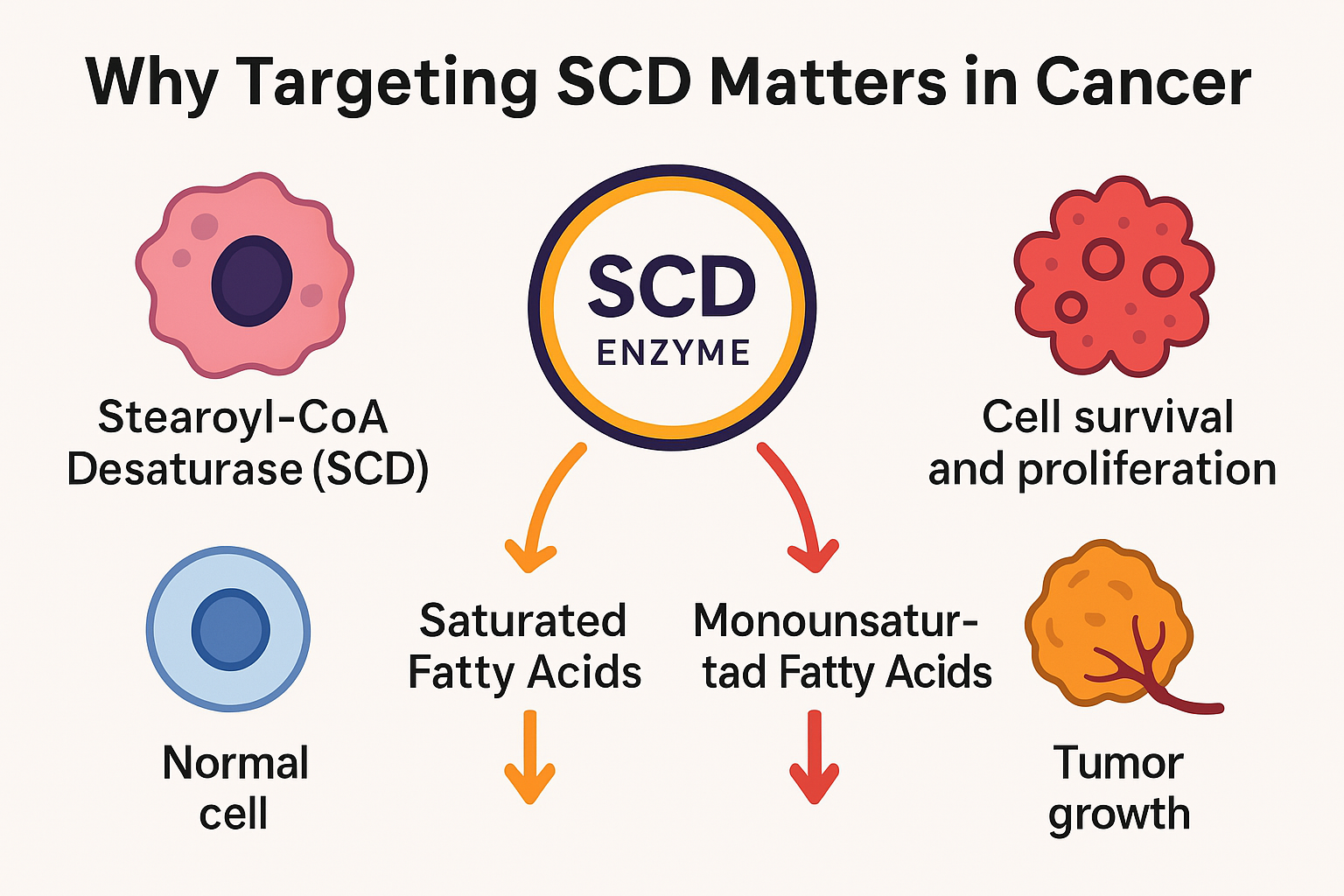
Figure 1. The Role of SCD in Cancer Progression
A recent study has made a breakthrough in this direction by discovering irreversible SCD inhibitors with tumor selectivity. These compounds covalently bind to the enzyme’s active site, shutting down its activity specifically in cancerous contexts. Such an approach represents a promising strategy in precision oncology, where treatments are tailored not just to genetic profiles but also to unique metabolic vulnerabilities of tumors.
The discovery of tumor-specific SCD inhibitors marks a shift in cancer drug development—one that targets the metabolic lifelines of cancer while minimizing harm to the patient.
Why Targeting SCD Matters in Cancer
As cancer research increasingly focuses on the metabolic dependencies of tumor cells, Stearoyl-CoA Desaturase (SCD) has emerged as a promising target. SCD plays a critical role in fatty acid metabolism by catalyzing the conversion of saturated fatty acids into monounsaturated fatty acids (MUFAs), which are essential for maintaining membrane fluidity, signaling, and energy storage.
In cancer, SCD activity is often upregulated, providing tumor cells with a continuous supply of MUFAs that support rapid proliferation and resistance to cellular stress. Elevated SCD expression has been observed in various cancers, including breast, prostate, liver, and lung, and is frequently associated with poor prognosis and treatment resistance.
Targeting SCD in cancer therapy is attractive because it disrupts the metabolic adaptations that tumors rely on. However, SCD is also active in healthy tissues, such as the liver and adipose tissue, where it regulates lipid homeostasis. Inhibiting SCD systemically has shown efficacy in preclinical cancer models but has also been linked to side effects, including skin and eye toxicity due to impaired lipid synthesis in normal cells.
This therapeutic dilemma has driven the search for tumor-selective SCD inhibitors—compounds that can preferentially inhibit SCD in cancer cells without affecting its function in normal tissues. Achieving this selectivity is crucial for translating metabolic inhibition into safe, targeted therapies. Researchers believe that cancer cells’ altered membrane composition, stress response pathways, or redox states may provide opportunities for selective inhibition strategies.
By focusing on the unique metabolic profiles of cancer cells, scientists are working to exploit SCD as a vulnerability that can be turned into a therapeutic advantage. Understanding why and how cancer depends on SCD is a foundational step toward the development of safer, more precise anticancer agents.
The Discovery – Tumor-Specific Irreversible SCD Inhibitors
In a significant breakthrough, researchers have identified a new class of irreversible inhibitors that selectively target Stearoyl-CoA Desaturase (SCD) in cancer cells. This innovation represents a novel approach to metabolic therapy, addressing the long-standing challenge of balancing efficacy with safety.
Using structure-based drug design and chemical screening, the study focused on modifying known SCD inhibitors—particularly MF-438 analogs—to enhance their selectivity and binding strength. The result was a series of covalent inhibitors that bind irreversibly to the enzyme’s active site, locking it in an inactive state. Among them, one compound stood out for its ability to preferentially inhibit SCD in tumor cells while sparing normal cells.
What sets these compounds apart is their tumor specificity. Unlike conventional SCD inhibitors that affect both cancerous and healthy tissues, the new analogs were engineered to exploit differences in the tumor microenvironment, such as altered lipid composition, enzyme conformation, or redox states. These conditions may make the covalent inhibitors more reactive in tumor cells, enhancing their selective potency.
Cell viability assays revealed that the new inhibitors significantly suppressed proliferation in multiple cancer cell lines while showing minimal cytotoxicity in non-cancerous counterparts. Proteomic profiling further confirmed that the irreversible inhibitors covalently modified the target protein in tumor cells, validating their mechanism of action.
This discovery offers a promising pathway toward precision metabolic therapy. Irreversible inhibition not only increases potency and duration of action but may also reduce dosing frequency and off-target effects. The ability to tailor such compounds for tumor-specific conditions could pave the way for a new generation of safer, more effective anticancer drugs.
By bridging medicinal chemistry and tumor biology, this research brings us closer to selectively disarming cancer’s metabolic engines—without collateral damage.
Mechanism of Action and Preclinical Potential
The newly discovered tumor-specific irreversible SCD inhibitors work through a sophisticated mechanism that enhances both their potency and selectivity. Unlike reversible inhibitors, which bind temporarily, these compounds form a covalent bond with Stearoyl-CoA Desaturase (SCD), effectively locking the enzyme in an inactive state. This irreversible mechanism not only improves inhibition durability but also reduces the likelihood of off-target effects by requiring precise molecular alignment—conditions more commonly found in cancer cells.
The covalent binding occurs at a nucleophilic site within the enzyme’s active region, and the chemical structure of the inhibitors was designed to exploit conformational differences between the SCD found in tumor versus normal cells. The cancer microenvironment often presents with altered lipid composition, oxidative stress, or structural variations that make SCD more susceptible to covalent targeting. This tumor-biased reactivity is a key to the inhibitors’ selective cytotoxicity.
In preclinical studies, the lead compounds demonstrated significant inhibition of tumor cell proliferation across various cancer cell lines, including those with high SCD expression. Importantly, they showed minimal effects on non-cancerous cells, a major advantage over traditional SCD inhibitors known for systemic toxicity.
Furthermore, mass spectrometry-based proteomic profiling confirmed that the inhibitors covalently modified SCD in situ, validating the mechanism of action at the molecular level. The irreversible inhibition also correlated with long-lasting effects on lipid metabolism and cancer cell viability, suggesting potential for lower dosing and reduced treatment frequency.
These findings mark a crucial step forward in cancer drug development. By leveraging irreversible, tumor-specific inhibition, researchers are not only refining metabolic targeting strategies but also setting the stage for clinical translation. The next phase will likely focus on in vivo efficacy and safety profiles, but the preclinical promise is already strong.
What This Means for Future Cancer Drug Development
The development of tumor-specific irreversible inhibitors of Stearoyl-CoA Desaturase (SCD) signifies more than a technical advancement—it represents a paradigm shift in how cancer drugs are conceived and designed. As researchers move beyond traditional cytotoxic agents and toward targeting metabolic dependencies, precision becomes the name of the game.
The approach detailed in this study leverages cancer cells’ altered metabolism and unique microenvironment to achieve selective inhibition, reducing toxicity and improving therapeutic windows. This aligns with the broader movement toward precision oncology, where treatments are tailored not only to genetic mutations but also to metabolic and biochemical signatures.
This breakthrough opens several doors. First, it validates irreversible inhibition as a viable and potentially superior strategy for targeting metabolic enzymes in cancer. Historically, covalent inhibitors were avoided due to concerns about off-target effects. However, recent successes—such as covalent EGFR and BTK inhibitors—have reignited interest in this approach. The current findings reinforce that, with proper design, covalent inhibitors can be both selective and safe.
Second, it highlights the untapped potential of tumor metabolism as a rich source of druggable vulnerabilities. As more cancers are found to depend on fatty acid synthesis and desaturation, metabolic targeting strategies like this may become standard components of combination therapies.
Finally, this study sets a framework for developing next-generation inhibitors—not just for SCD but for a host of enzymes with differential activity in tumor versus normal tissues. By integrating structure-based drug design, proteomics, and tumor biology, the field is poised to deliver smarter, safer drugs.
In short, this research represents a new frontier: targeted metabolic therapy with precision, durability, and reduced toxicity—a promising vision for the future of cancer treatment.
References
Igal, R. A. (2016). Roles of StearoylCoA desaturase-1 in the regulation of cancer cell growth, survival and tumorigenesis. Cancers, 8(3), 30. https://doi.org/10.3390/cancers8030030
Li, J., Gu, D., Lee, S. S.-Y., Song, B., & Bandyopadhyay, S. (2021). Discovery of tumor-specific irreversible inhibitors of stearoyl coenzyme A desaturase. International Journal of Molecular Sciences, 22(5), 1742. https://doi.org/10.3390/ijms22051742
Röhrig, F., & Schulze, A. (2016). The multifaceted roles of fatty acid synthesis in cancer. Nature Reviews Cancer, 16(11), 732–749. https://doi.org/10.1038/nrc.2016.89
Currie, E., Schulze, A., Zechner, R., Walther, T. C., & Farese, R. V. (2013). Cellular fatty acid metabolism and cancer. Cell Metabolism, 18(2), 153–161. https://doi.org/10.1016/j.cmet.2013.05.017
Bederman, I. R., Fiehn, O., & Kurland, I. J. (2015). Evidence for a tumor-specific metabolic signature in cancer. Cancer & Metabolism, 3(1), 6. https://doi.org/10.1186/s40170-015-0133-y
Igal, R. A. (2011). Stearoyl CoA desaturase-1: New insights into a central regulator of cancer metabolism. Biochimica et Biophysica Acta (BBA) – Molecular and Cell Biology of Lipids, 1811(11), 1055–1060. https://doi.org/10.1016/j.bbalip.2011.07.008
Roongta, U. V., Pabalan, J. G., Wang, X., Ryseck, R.-P., Fargnoli, J., Henley, B. J., … & Bence, K. K. (2011). Cancer cell dependence on unsaturated fatty acids implicates stearoyl-CoA desaturase as a target for cancer therapy. Molecular Cancer Research, 9(11), 1551–1561. https://doi.org/10.1158/1541-7786.MCR-11-0264
Scaglia, N., & Igal, R. A. (2008). Inhibition of stearoyl-CoA desaturase 1 expression in human lung adenocarcinoma cells impairs tumorigenesis. International Journal of Oncology, 33(4), 839–850. https://doi.org/10.3892/ijo_00000062
Wang, C., Falany, C. N., & Leyens, G. (2017). Targeting metabolic pathways to treat cancer: Emerging roles of stearoyl-CoA desaturase inhibitors. Frontiers in Oncology, 7, 106. https://doi.org/10.3389/fonc.2017.00106
Grimm, M. O., Kuchenbecker, J., & Grosgen, S. (2016). From brain to cell membrane: The role of lipid metabolism in Alzheimer’s disease and cancer. Molecular Neurobiology, 53(3), 1356–1373. https://doi.org/10.1007/s12035-015-9098-6
Mahdaviani, K., & Nomura, D. K. (2020). Covalent chemical proteomics: A strategy to discover functional and selective covalent probes. Cell Chemical Biology, 27(4), 428–444. https://doi.org/10.1016/j.chembiol.2020.03.002
Nomura, D. K., & Cravatt, B. F. (2012). Activity-based protein profiling for biochemical pathway discovery in cancer. Nature Reviews Cancer, 12(12), 825–837. https://doi.org/10.1038/nrc3391
Puri, P., Baillie, R. A., Wiest, M. M., Mirshahi, F., Choudhury, J., Cheung, O., … & Sanyal, A. J. (2007). A lipidomic analysis of nonalcoholic fatty liver disease. Hepatology, 46(4), 1081–1090. https://doi.org/10.1002/hep.21763
Singh, J., Petter, R. C., Baillie, T. A., & Whitty, A. (2011). The resurgence of covalent drugs. Nature Reviews Drug Discovery, 10(4), 307–317. https://doi.org/10.1038/nrd3410
Vander Heiden, M. G., & DeBerardinis, R. J. (2017). Understanding the intersections between metabolism and cancer biology. Cell, 168(4), 657–669. https://doi.org/10.1016/j.cell.2016.12.039
Harten, S. K., Esteban, M. A., Shukla, D., Ashcroft, M., & Maxwell, P. H. (2010). Inactivation of the FIH hydroxylase allows tumor growth in vivo. Proceedings of the National Academy of Sciences, 107(43), 19700–19705. https://doi.org/10.1073/pnas.1007473107
Cheng, Z., & Li, M. (2020). Targeting tumor-specific metabolic pathways: A promising therapeutic strategy for cancer. International Journal of Molecular Sciences, 21(20), 7605. https://doi.org/10.3390/ijms21207605